Stay updated with expert articles, tips, and stories that inspire and inform
-
Read more ...
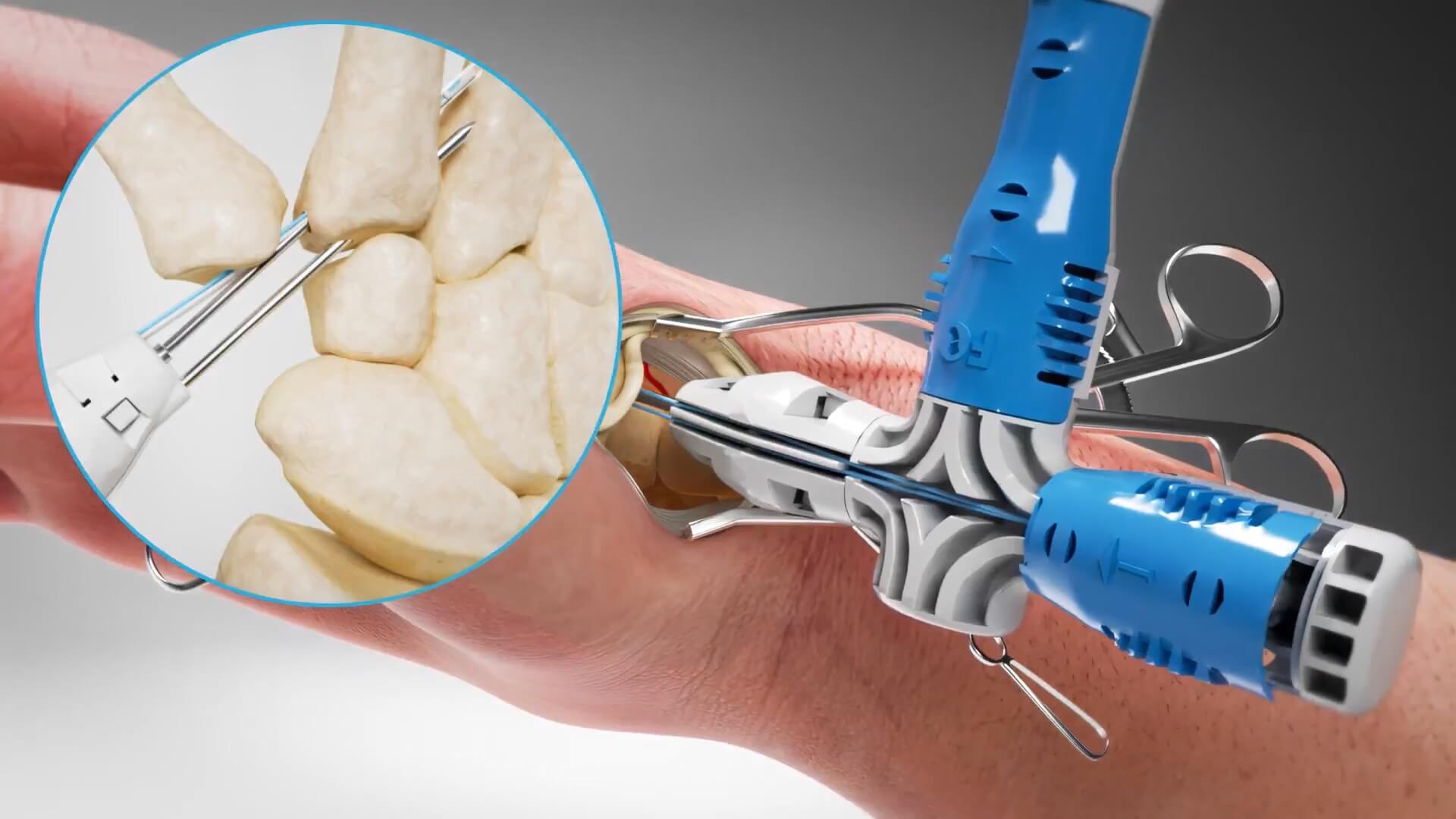
Field Orthopaedics is a medical technology company dedicated to advancing the field of hand surgery through innovation. They specialize in the development of cutting-edge medical implants and surgical solutions that improve outcomes for both patients and surgeons and transform the standard of care in orthopaedic hand procedures. Challenge 3D animation for the demonstration of a
-
October 12, 2024Read more ...
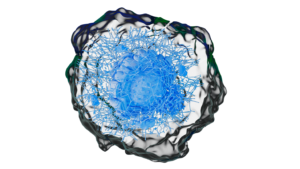
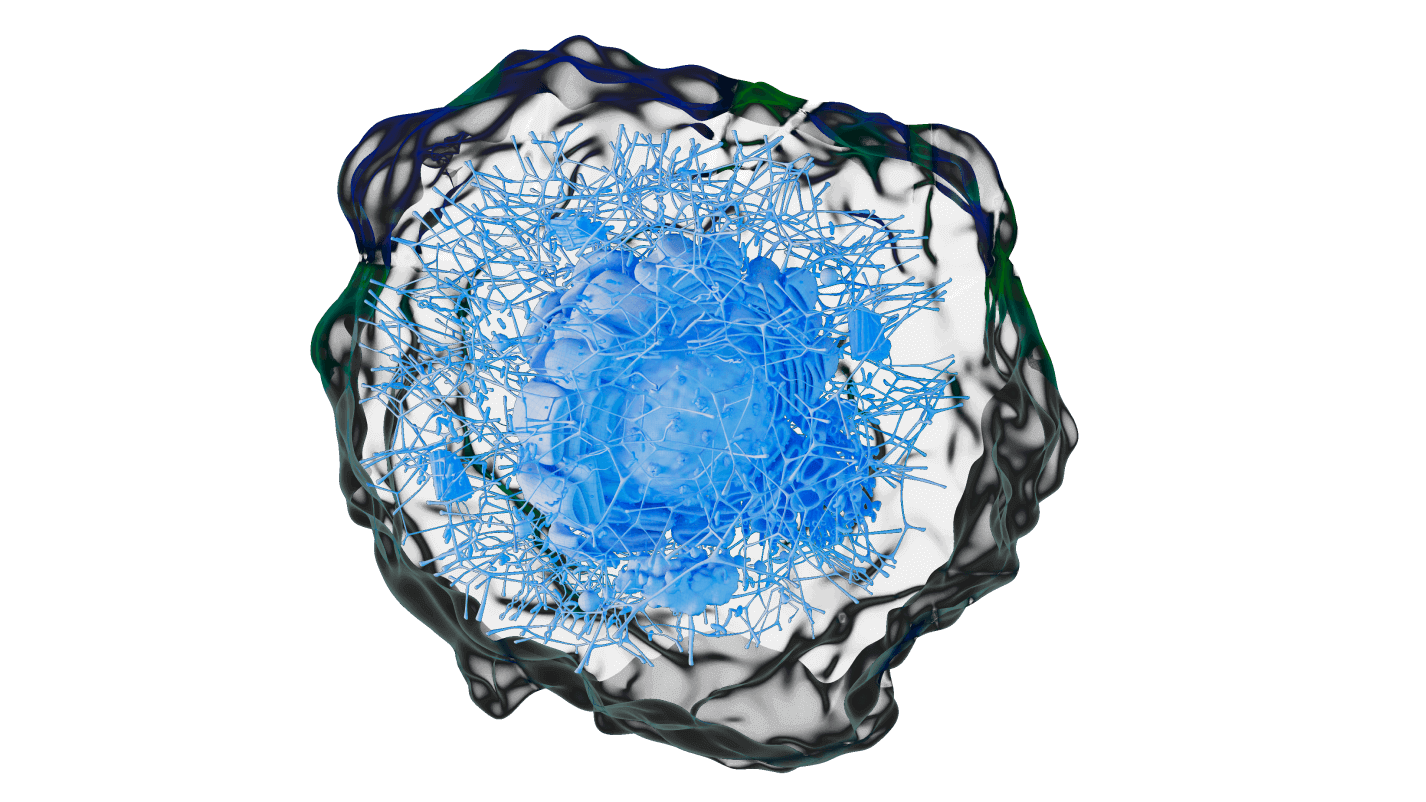
Project scope To showcase expertise in an integrated robotic platform for tissue engineering and developing artificial joints using stem cells and 3D printing. KU Leuven is an international community where innovative research forms the basis of all academic programs. JOINTPROMISE project paves the way for high-volume, affordable production of entire biological joints, addressing a major
-
June 18, 2024Read more ...
We are thrilled to announce that our 3D animation video for Agnucaston, created for Bionorica SE, has been named a finalist for the COMPRIX Kreative Healthcare Kommunikation 2024 awards! Being recognized among the best in the industry is an incredible honor, and we are eagerly looking forward to the awards ceremony in September to find
-
June 3, 2024Read more ...
c-LEcta is the world-leading industrial biotechnology company supplying best-in-class engineered enzymes to regulated industries. Goal: to inform existing customers & leads about the new product, its features, and advantages. In this project, we created a 2D custom animation to help c-LEcta inform their customers and leads about their new product, its features, and advantages.
-
May 13, 2024Read more ...
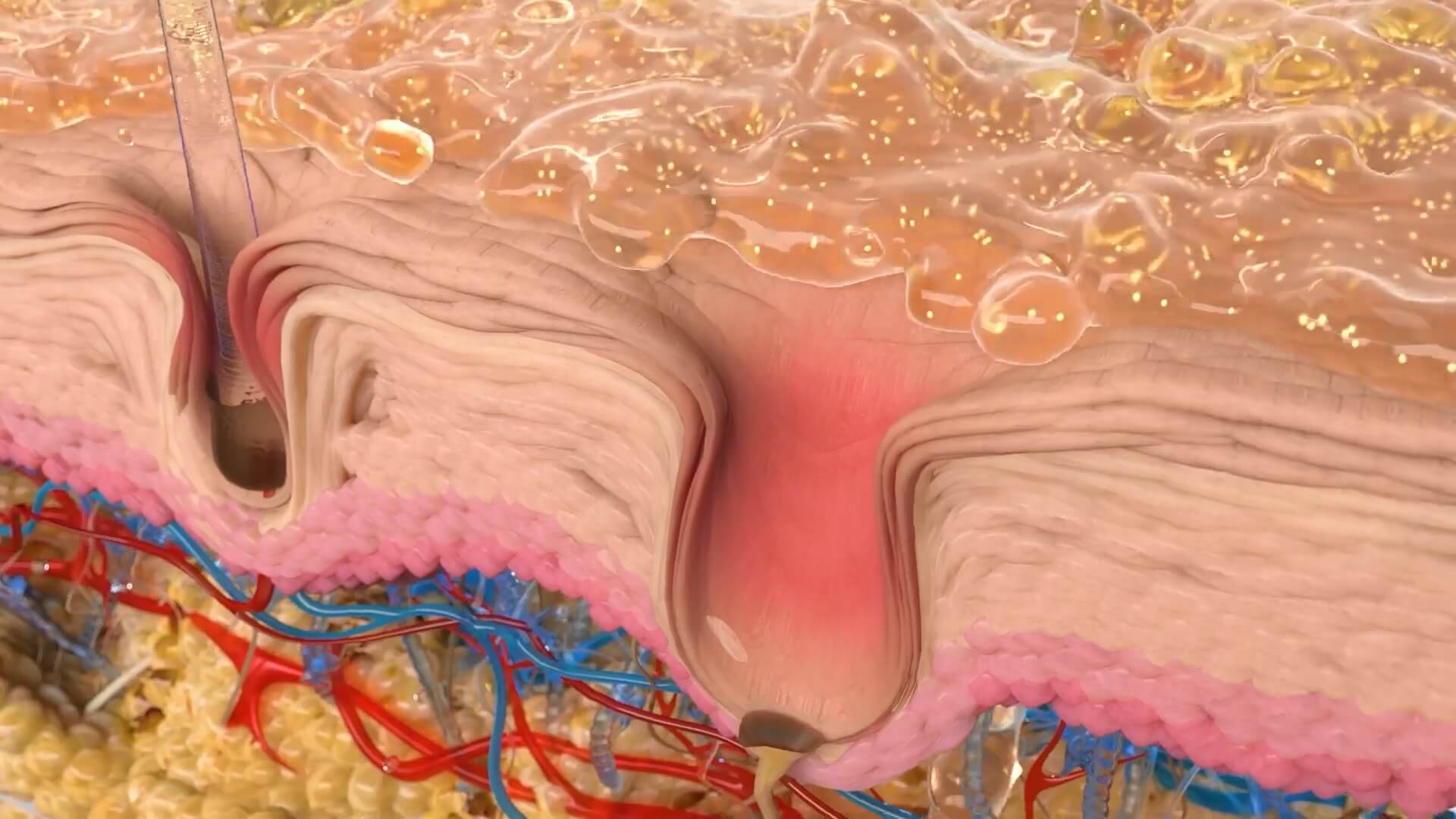
Aesthetic medicine companies have to appeal to two distinctively different types of audiences. First, they need to communicate with end consumers of beauty products. These consumers often lack the necessary knowledge about the anatomy and biochemistry of skin, beauty products, and beauty procedures. They need an education that will simplify complex terms and processes and